Verification of candidate biomarkers requires particular assays to selectively detect and quantify goal proteins in accessible biofluids.
The major goal of verification is to display screen potential biomarkers to make sure that solely the very best high quality candidates from the invention part are taken ahead into preclinical validation.
Because antibody <em>reagents</em> for a scientific <em>grade</em> immunoassay typically exist for a small quantity of candidates, various methodologies are required to credential new and unproven candidates in a statistically viable quantity of serum or plasma samples.
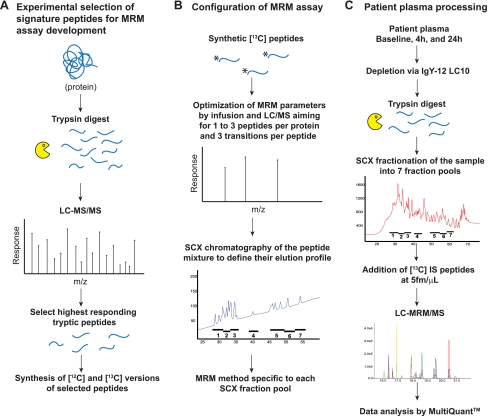
Using a number of response monitoring coupled with stable isotope dilution MS, we developed quantitative, multiplexed assays in plasma for six proteins of scientific relevance to cardiac harm.
The course of described doesn’t require antibodies for immunoaffinity enrichment of both proteins or peptides. Limits of detection and quantitation for every signature peptide used as surrogates for the goal proteins have been decided by the strategy of normal addition utilizing artificial peptides and plasma from a wholesome donor.
Limits of quantitation ranged from 2 to 15 ng/ml for many of the goal proteins. Quantitative measurements have been obtained for one to 2 signature peptides derived from every goal protein, together with low abundance protein markers of cardiac harm in the nanogram/milliliter vary such because the cardiac troponins. Intra- and interassay coefficients of variation have been predominantly <10 and 25%, respectively.
The configured multiplex assay was then used to measure ranges of these proteins throughout three time factors in six sufferers present process alcohol septal ablation for hypertrophic obstructive cardiomyopathy.
These outcomes are the primary demonstration of a multiplexed, MS-based assay for detection and quantification of modifications in focus of proteins related to cardiac harm in the low nanogram/milliliter vary.
Our outcomes additionally exhibit that these assays retain the mandatory precision, reproducibility, and sensitivity to be utilized to novel and uncharacterized candidate biomarkers for verification of proteins in blood.
Subsecond adsorption and desorption of dopamine at carbon-fiber microelectrodes.
High-repetition fast-scan cyclic voltammetry and chronoamperometry have been used to quantify and characterize the kinetics of dopamine and dopamine-o-quinone adsorption and desorption at carbon-fiber microelectrodes.
A stream injection evaluation system was used for the exact introduction and elimination of a bolus of electroactive substance on a sub-second time scale to the disk-shaped floor of a microelectrode that was fabricated from a single carbon fiber (Thornel kind T650 or P55).
Pretreatment of the electrode surfaces consisted of soaking them in purified isopropyl alcohol for a minimal of 10 min, which resulted in S/N rising by 200-400% for dopamine above that for people who have been soaked in reagent grade solvent.
Because of adsorption, excessive scan charges (2,000 V/s) are proven to exhibit equal S/N ratios as in comparison with slower, extra conventional scan charges. In addition, the steady-state response to a focus bolus is proven to happen extra quickly when cyclic voltammetric scans are repeated at brief intervals (four ms).
The new methodologies enable for extra correct determinations of the kinetics of neurotransmitter launch occasions (10-500 ms) in organic methods.
Brain slice and in vivo experiments utilizing T650 cylinder microelectrodes present that voltammetrically measured uptake kinetics in the caudate are quicker utilizing 2,000 V/s and 240 Hz measurements, as in comparison with 300 V/s and 10 Hz.