Product name
Androgen receptor (AR) ELISA kit
Full product name
Human Androgen Receptor (AR) ELISA Kit
Product Synonym Names
SIA; DHTR; HUMARA; K.D.; NR3C4; SBMA; SMAX1; TFM; dihydrotestosterone receptor; Nuclear Receptor Subfamily 3, Group C, Member 4; Testicular Feminization; Kennedy’s disease
Product gene name: AR Elisa kit
OMIM: 300068
3D structure: ModBase 3D Structure for P10275
Species reactivity: Human
Specificity: This assay has high sensitivity and excellent specificity for the detection of RA. No significant cross-reactivity or interference was observed between Elisa-and-PCR-kits-AR and analogues.
Samples
Homogenates of human tissues, cell lysates, and other biological fluids
Test type: Quantitative sandwich
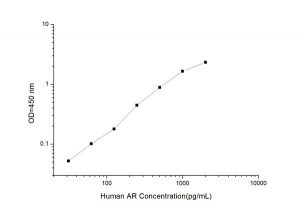
Detection range: 0.312-20ng/mL
Sensitivity: <0.118ng/mL
Intra-assay precision
Intra-assay Precision (Precision Within an Assay): 3 samples with low, medium and high-level RA were tested 20 times on one plate, respectively. Intra-assay: CV<10%
Inter-assay precision
Inter-assay Precision (Inter-Assay Precision): 3 samples with low, medium and high-level RA were tested on 3 different plates, 8 replicates on each plate. CV(%) = SD/meanX100. Inter-assay: CV<12%
Preparation and Storage
The stability of the kit is determined by the rate of loss of activity. The loss rate of this kit is less than 5% within the expiration date under proper storage conditions. To minimize the further influence on performance, operating procedures and laboratory conditions, especially room temperature, air humidity, and incubator temperature, should be strictly controlled. It is also strongly recommended that the entire test be performed by the same operator from start to finish.
ISO certification
Manufactured in an ISO 9001:2008 and ISO 13485:2003 certified laboratory.
Supply Chain Verification
Manufactured in a laboratory with traceable raw materials. Bulk orders can normally be prepared to customer specifications, please inquire.
Product note
Information selected from the online data sheet is extracted from bioinformatics databases, sometimes resulting in the ambiguous or non-relevant product information. It is the customer’s responsibility to review, verify and evaluate the information to ensure it matches their requirements prior to purchasing the kit. Our ELISA Kit assays are dynamic research tools and may be updated and improved from time to time.
If the format of this assay is important to you, please request the current manual or contact our technical support team with a presales inquiry before placing an order. We will confirm the current trial details. We cannot guarantee that the sample manual posted online is the most up-to-date manual, it is intended to serve as an example only. Consult the instructions for use provided with the assay kit for precise details.
Other notes
On occasion, small volumes of AR Elisa kit vials may become trapped in the product vial seal during shipping and storage. If necessary, briefly spin the vial in a tabletop centrifuge to dislodge any liquid in the vial cap. Certain products may require dry ice shipping and an additional dry ice fee may apply.
Search Terms for AR Purchase
MBS2022364 is a ready-to-use microwell and strip plate ELISA (Enzyme-Linked Immunosorbent Assay) Kit for testing the presence of Androgen Receptor (AR) ELISA Kit target analytes in biological samples. The concentration gradients of the kit standards or positive controls generate a theoretical detection range of the kit in biological research samples containing AR.
The MBS2022364 ELISA analytical biochemical technique is based on AR antigen-AR antibody (immunosorbent) interactions and an HRP colourimetric detection system to detect AR antigen targets in samples. The ELISA Kit is designed to detect native, non-recombinant AR. Appropriate sample types may include undiluted body fluids and/or tissue homogenates, secretions. Quality control assays that evaluated reproducibility identified intra-assay CV (%) and inter-assay CV (%).
Product related information for ar kit Elisa
Intended Uses: The kit is a sandwich enzyme immunoassay for the in vitro quantitative measurement of AR in human tissue homogenates, cell lysates, and other biological fluids.
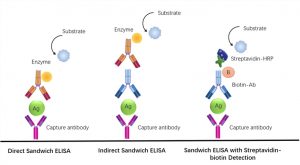
Assay Principle: The microplate provided in this kit has been previously coated with an RA-specific antibody. Standards or samples are then added to the appropriate microplate wells with a biotin-conjugated antibody specific for RA. Avidin conjugated with horseradish peroxidase (HRP) is then added to each well of the microplate and incubated.
After adding the TMB Substrate Solution, only wells containing RA, Biotin-Conjugated Antibody, and Enzyme-Conjugated Avidin will exhibit a colour change. The enzyme-substrate reaction is terminated by the addition of sulfuric acid solution and the colour change is measured spectrophotometrically at a wavelength of 450nm +/- 10nm. The concentration of AR in the samples is then determined by comparing the O.D. of the samples to the standard curve.
Precautions
All MyBioSource products are for scientific laboratory research purposes and not for diagnostic, therapeutic, prophylactic, or in vivo use. Through your purchase, you expressly represent and warrant to MyBioSource that you will properly test and use any Product purchased from MyBioSource in accordance with industry standards. MyBioSource and its authorized distributors reserve the right to refuse to process any order where we reasonably believe the intended use will not meet our acceptable guidelines.
Disclaimer
While every effort has been made to ensure the accuracy of the information provided in this datasheet, MyBioSource shall not be responsible for any omissions or errors contained in this document. MyBioSource reserves the right to make changes to this datasheet at any time without notice.