An unmet want in cell engineering is the provision of a single transgene encoded, functionally inert, human polypeptide that may serve a number of functions, together with ex vivo cell choice, in vivo cell monitoring, and as a goal for in vivo cell ablation.
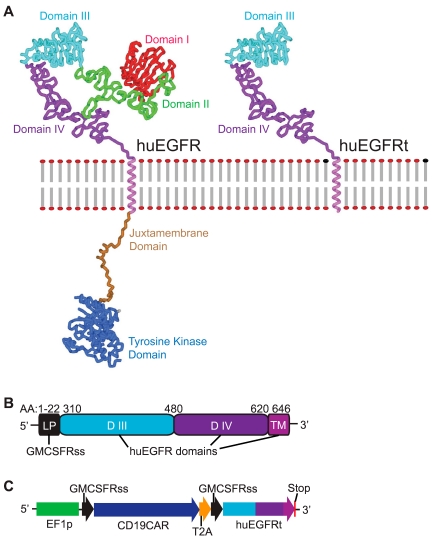
Here we describe a truncated human EGFR polypeptide (huEGFRt) that’s devoid of extracellular N-terminal ligand binding domains and intracellular receptor tyrosine kinase exercise however retains the native amino acid sequence, kind I transmembrane cell surface localization, and a conformationally intact binding epitope for pharmaceutical-grade anti-EGFR monoclonal antibody, cetuximab (Erbitux).
After lentiviral transduction of human T cells with vectors that coordinately specific tumor-specific chimeric antigen receptors and huEGFRt, we present that huEGFRt serves as a extremely environment friendly choice epitope for chimeric antigen receptor(+) T cells utilizing biotinylated cetuximab in conjunction with present good manufacturing practices (cGMP)-grade anti-biotin immunomagnetic microbeads.
Moreover, huEGFRt supplies a cell surface marker for in vivo monitoring of adoptively transferred T cells utilizing each move cytometry and immunohistochemistry, and a goal for cetuximab-mediated antibody-dependent mobile cytotoxicity and in vivo elimination.
The versatility of huEGFRt and the provision of pharmaceutical-grade reagents for its scientific utility denote huEGFRt as a big new instrument for mobile engineering.
Localization of human BRCA1 and its loss in high-grade, non-inherited breast carcinomas.
Although the hyperlink between the BRCA1 tumour-suppressor gene and hereditary breast and ovarian most cancers is established, the function, if any, of BRCA1 in non-familial cancers is unclear.
BRCA1 mutations are uncommon in sporadic cancers, however loss of BRCA1 ensuing from decreased expression or incorrect subcellular localization is postulated to be vital in non-familial breast and ovarian cancers.
Epigenetic loss, nevertheless, has not obtained basic acceptance resulting from controversy relating to the subcellular localization of BRCA1 proteins, studies of which have ranged from completely nuclear, to conditionally nuclear, to the ER/golgi, to cytoplasmic invaginations into the nucleus. In an try and resolve this situation, we have now comprehensively characterised 19 anti-BRCA1 antibodies.
These reagents detect a 220-kD protein localized in discrete nuclear foci in all epithelial cell strains, together with these derived from breast malignancies. Immunohistochemical staining of human breast specimens additionally revealed BRCA1 nuclear foci in benign breast, invasive lobular cancers and low-grade ductal carcinomas.
Conversely, BRCA1 expression was decreased or undetectable in the bulk of high-grade, ductal carcinomas, suggesting that absence of BRCA1 could contribute to the pathogenesis of a big proportion of sporadic breast cancers.